Medical expert of the article
New publications
Parathyroid glands
Last reviewed: 04.07.2025

All iLive content is medically reviewed or fact checked to ensure as much factual accuracy as possible.
We have strict sourcing guidelines and only link to reputable media sites, academic research institutions and, whenever possible, medically peer reviewed studies. Note that the numbers in parentheses ([1], [2], etc.) are clickable links to these studies.
If you feel that any of our content is inaccurate, out-of-date, or otherwise questionable, please select it and press Ctrl + Enter.
In 1879, Swedish scientist S. Sandstrom described the parathyroid glands in humans and gave them a name. The parathyroid glands are vital organs. Their function is to produce and secrete parathyroid hormone (PTH), one of the main regulators of calcium and phosphorus metabolism.
The paired superior parathyroid gland (glandula parathyroidea superior) and inferior parathyroid gland (glandula parathyroidea inferior) are round or ovoid bodies located on the back surface of each of the lobes of the thyroid gland: one gland at the top, the other at the bottom. The length of each gland is 4-8 mm, the width is 3-4 mm, the thickness is 2-3 mm. The number of these glands is not constant and can vary from 2 to 7-8, on average there are four. The total mass of the glands is on average 1.18 g.
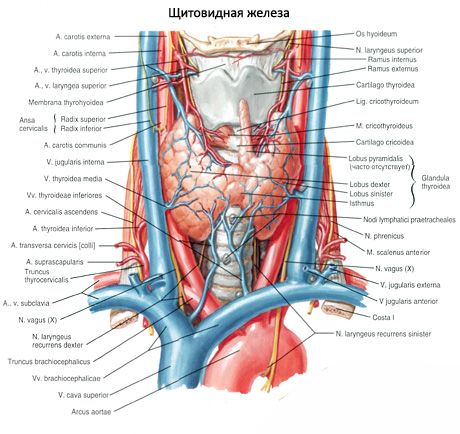
The parathyroid glands differ from the thyroid gland in their lighter color (in children they are pale pink, in adults - yellowish-brown). Often the parathyroid glands are located at the site of penetration of the thyroid tissue by the inferior thyroid arteries or their branches. The parathyroid glands are separated from the surrounding tissues by their own fibrous capsule, from which connective tissue layers extend into the glands. The latter contain a large number of blood vessels and divide the parathyroid glands into groups of epithelial cells.
The parenchyma of the glands is formed by the main and acidophilic parathyrocytes, which form cords and clusters surrounded by thin bundles of connective tissue fibers. Both types of cells are considered as different stages of parathyrocyte development. The main parathyrocytes have a polyhedral shape, basophilic cytoplasm with a large number of ribosomes. Among these cells, dark (actively secreting) and light (less active) are distinguished. Acidophilic parathyrocytes are large, with clear contours, contain many small mitochondria with glycogen particles.
The hormone of the parathyroid glands, parathyroxine (parathyroid hormone), is protein in composition and is involved in the regulation of phosphorus-calcium metabolism. Parathyroid hormone reduces calcium excretion in urine and increases its absorption in the intestine in the presence of vitamin D. Thyrocalcitonin is an antagonist of parathyroid hormone.
Embryogenesis of the parathyroid glands
The parathyroid glands develop from the epithelium of the paired III and IV gill pockets. In the 7th week of development, the epithelial rudiments of the bodies separate from the walls of the gill pockets and, in the process of growth, move in the caudal direction. Subsequently, the forming parathyroid glands occupy a permanent position for them on the posterior surfaces of the right and left lobes of the thyroid gland.
 [ 7 ], [ 8 ], [ 9 ], [ 10 ], [ 11 ], [ 12 ]
[ 7 ], [ 8 ], [ 9 ], [ 10 ], [ 11 ], [ 12 ]
Vessels and nerves of the parathyroid glands
The blood supply to the parathyroid glands is provided by branches of the superior and inferior thyroid arteries, as well as the esophageal and tracheal branches. Venous blood flows out through the veins of the same name. The innervation of the parathyroid glands is similar to the innervation of the thyroid gland.
Age-related features of the parathyroid glands
The total mass of the parathyroid glands in a newborn fluctuates between 6 and 9 mg. During the first year of life, their total mass increases 3-4 times, by the age of 5 it doubles again, and by the age of 10 it triples. After 20 years, the total mass of the four parathyroid glands reaches 120-140 mg and remains constant until old age. At all ages, the mass of the parathyroid glands in women is slightly greater than in men.
Usually a person has two pairs of parathyroid glands (upper and lower), located on the back surface of the thyroid gland, outside its capsule, near the upper and lower poles. However, the number and localization of the parathyroid glands can vary; sometimes up to 12 parathyroid glands are found. They can be located in the tissue of the thyroid and thymus glands, in the anterior and posterior mediastinum, in the pericardium, behind the esophagus, in the area of the bifurcation of the carotid artery. The upper parathyroid glands have the shape of a flattened ovoid, the lower ones are spherical. Their dimensions are approximately from 6x3 to 4x1.5 - 3 mm, the total weight is from 0.05 to 0.5 g, the color is reddish or yellowish-brown. Blood supply to the parathyroid glands is carried out mainly by the branches of the inferior thyroid artery, venous outflow occurs through the veins of the thyroid gland, esophagus and trachea. The parathyroid glands are innervated by sympathetic fibers of the recurrent and superior laryngeal nerves, parasympathetic innervation is carried out by the vagus nerves. The parathyroid glands are covered with a thin connective tissue capsule; the septa extending from it penetrate into the glands. They contain blood vessels and nerve fibers. The parenchyma of the parathyroid glands consists of parathyrocytes, or chief cells, among which, according to the degree of staining, hormonally active light or shiny cells, as well as resting dark cells, are distinguished. The chief cells form clusters, cords and clusters, and in the elderly - follicles with colloid in the cavity. In adults, cells appear, located mainly along the periphery of the parathyroid glands, stained with eosin - eosinophilic or oxyphilic cells, which are degenerating chief cells. In the parathyroid glands, transitional forms between the principal and oxyphilic cells can also be found.
The first successes in clarifying the issues of synthesis, decoding the structure, studying the metabolism of parathyroid hormone were achieved after 1972. Parathyroid hormone is a single-chain polypeptide consisting of 84 amino acid residues, devoid of cysteine, with a molecular weight of approximately 9500 daltons, formed in the parathyroid glands from a bioprecursor - proparathyroid hormone (proPTH), which has 6 additional amino acids at the NH 2 -end. ProPTH is synthesized in the main cells of the parathyroid glands (in their granular endoplasmic reticulum) and in the process of proteolytic cleavage in the Golgi apparatus is converted into parathyroid hormone. Its biological activity is significantly lower than the activity of PTH. Apparently, proPTH is absent in the blood of healthy people, but in pathological conditions (with adenoma of the parathyroid glands) it can secrete into the blood along with PTH. Recently, a precursor of proPTH, preproPTH, was discovered that contains an additional 25 amino acid residues at the NH2-terminus. Thus, preproPTH contains 115 amino acid residues, proPTH - 90, and PTH - 84.
The structure of bovine and porcine parathyroid hormone has now been fully established. Human parathyroid hormone has been isolated from parathyroid gland adenomas, but its structure has only been partially deciphered. There are differences in the structure of parathyroid hormone, but animal and human parathyroid hormones exhibit cross-immunoreactivity. The polypeptide consisting of the first 34 amino acid residues practically retains the biological activity of the natural hormone. This allows us to assume that the remaining almost % of the molecule at the carboxyl end are not directly related to the main effects of parathyroid hormone. Fragment 1-29 of parathyroid hormone also exhibits a certain biological and immunological activity. Biologically inactive fragment 53-84 also exhibits an immunological effect, i.e. these properties of parathyroid hormone are exhibited by at least 2 sections of its molecule.
The parathyroid hormone circulating in the blood is heterogeneous and differs from the native hormone secreted by the parathyroid glands. At least three different types of parathyroid hormone are distinguished in the blood: intact parathyroid hormone with a molecular weight of 9,500 daltons; biologically inactive substances from the carboxyl part of the parathyroid hormone molecule with a molecular weight of 7,000-7,500 daltons; biologically active substances with a molecular weight of about 4,000 daltons.
Even smaller fragments were found in venous blood, indicating that they were formed on the periphery. The main organs where parathyroid hormone fragments were formed were the liver and kidneys. Fragmentation of parathyroid hormone in these organs was increased in liver pathology and chronic renal failure (CRF). Under these conditions, parathyroid hormone fragments remained in the bloodstream significantly longer than in healthy individuals. The liver absorbed mainly intact parathyroid hormone, but did not remove either carboxyl-terminal or amino-terminal parathyroid hormone fragments from the blood. The kidneys played a leading role in parathyroid hormone metabolism. They accounted for almost 60% of the metabolic clearance of carboxyl-terminal immunoreactive hormone and 45% of the amino-terminal fragment of parathyroid hormone. The main area of metabolism of the active amino-terminal fragment of parathyroid hormone was bones.
Pulsating secretion of parathyroid hormone was detected, most intense at night. After 3-4 hours from the beginning of night sleep, its content in the blood is 2.5-3 times higher than the average daytime level.
The main function of parathyroid hormone is to maintain calcium homeostasis. At the same time, serum calcium (total and especially ionized) is the main regulator of parathyroid hormone secretion (a decrease in calcium levels stimulates parathyroid hormone secretion, while an increase suppresses it), i.e., regulation is carried out according to the feedback principle. Under conditions of hypocalcemia, the conversion of proPTH into parathyroid hormone increases. The magnesium content in the blood also plays an important role in the release of parathyroid hormone (its increased level stimulates, while its decreased level suppresses parathyroid hormone secretion). The main targets of parathyroid hormone are the kidneys and skeletal bones, but the effect of parathyroid hormone on calcium absorption in the intestine, carbohydrate tolerance, serum lipid levels, its role in the development of impotence, skin itching, etc. is known.
To characterize the effect of parathyroid hormone on bones, it is necessary to provide brief information about the structure of bone tissue, the features of its physiological resorption and remodeling.
It is known that the bulk of calcium in the body (up to 99%) is contained in bone tissue. Since it is found in bone in the form of phosphorus-calcium compounds, % of the total phosphorus content is also found in bones. Their tissue, despite its apparent static nature, is constantly remodeled, actively vascularized and has high mechanical properties. Bone is a dynamic "depot" of phosphorus, magnesium and other compounds necessary to maintain homeostasis in mineral metabolism. Its structure includes dense mineral components that are closely related to the organic matrix, which consists of 90-95% collagen, small amounts of mucopolysaccharides and non-collagen proteins. The mineral part of the bone consists of hydroxyapatite - its empirical formula is Ca10 (PO4) 6 (OH) 2 - and amorphous calcium phosphate.
Bone is formed by osteoblasts originating from undifferentiated mesenchyme cells. These are mononuclear cells involved in the synthesis of components of the organic bone matrix. They are located in a monolayer on the bone surface and are in close contact with osteoid. Osteoblasts are responsible for the deposition of osteoid and its subsequent mineralization. The product of their vital activity is alkaline phosphatase, the content of which in the blood is an indirect indicator of their activity. Surrounded by mineralized osteoid, some osteoblasts turn into osteocytes - mononuclear cells, the cytoplasm of which forms canals associated with the canals of neighboring osteocytes. They do not participate in bone remodeling, but are involved in the process of perilacunar destruction, which is important for the rapid regulation of calcium levels in the blood serum. Bone resorption is carried out by osteoclasts - giant polynuclear cells, apparently formed by the fusion of mononuclear macrophages. It is also assumed that the precursors of osteoclasts may be hematopoietic stem cells of the bone marrow. They are mobile, form a layer in contact with the bone, located in areas of its greatest resorption. Due to the release of proteolytic enzymes and acid phosphatase, osteoclasts cause collagen degradation, hydroxyapatite destruction and the removal of minerals from the matrix. Newly formed poorly mineralized bone tissue (osteoid) is resistant to osteoclastic resorption. The functions of osteoblasts and osteoclasts are independent, but coordinated with each other, which leads to normal skeletal remodeling. Bone growth in length depends on endochondral ossification, growth in width and thickness - on periosteal ossification. Clinical studies with 47 Ca have shown that up to 18% of the total calcium content in the skeleton is renewed each year. When bones are damaged (fractures, infectious processes), the altered bone is resorbed and new bone is formed.
Cell complexes involved in the local process of bone resorption and formation are called basic multicellular remodeling units (BMUs). They regulate the local concentration of calcium, phosphorus and other ions, the synthesis of organic components of bone, in particular collagen, its organization and mineralization.
The main effect of parathyroid hormone in the bones of the skeleton is to enhance the resorption processes, affecting both mineral and organic components of the bone structure. Parathyroid hormone promotes the growth of osteoclasts and their activity, which is accompanied by an enhanced osteolytic effect and an increase in bone resorption. In this case, hydroxyapatite crystals dissolve with the release of calcium and phosphorus into the blood. This process is the main mechanism for increasing the level of calcium in the blood. It consists of three components: mobilization of calcium from the perilacunar bone (deep osteocytes); proliferation of osteoprogenerator cells into osteoclasts; maintaining a constant level of calcium in the blood by regulating its release from the bone (superficial osteocytes).
Thus, parathyroid hormone initially increases the activity of osteocytes and osteoclasts, enhancing osteolysis, causing an increase in the level of calcium in the blood and increased excretion of calcium and oxyproline in the urine. This is the first, qualitative, rapid effect of parathyroid hormone. The second effect of parathyroid hormone on bones is quantitative. It is associated with an increase in the pool of osteoclasts. With active osteolysis, a stimulus for increased proliferation of osteoblasts occurs, both resorption and bone formation with a predominance of resorption are activated. With an excess of parathyroid hormone, a negative bone balance occurs. This is accompanied by excessive excretion of oxyproline - a product of collagen degradation and sialic acids included in the structure of mucopolysaccharides. Parathyroid hormone activates cyclic adenosine monophosphate (cAMP). Increased excretion of cAMP in the urine after the administration of parathyroid hormone can serve as an indicator of tissue sensitivity to it.
The most important effect of parathyroid hormone on the kidneys is its ability to reduce phosphorus reabsorption, increasing phosphaturia. The mechanism of reduction in different parts of the nephron is different: in the proximal part, this effect of parathyroid hormone is due to increased permeability and occurs with the participation of cAMP, while in the distal part, it does not depend on cAMP. The phosphaturic effect of parathyroid hormone changes with vitamin D deficiency, metabolic acidosis, and decreased phosphorus content. Parathyroid hormones slightly increase the total tubular reabsorption of calcium. At the same time, it decreases it in the proximal parts and increases it in the distal parts. The latter is of dominant importance - parathyroid hormone decreases calcium clearance. Parathyroid hormone decreases the tubular reabsorption of sodium and its bicarbonate, which may explain the development of acidosis in hyperparathyroidism. It increases the formation of 1,25-dioxycholecalciferol 1,25(OH 2 )D 3 in the kidneys - the active form of vitamin D 3. This compound increases calcium reabsorption in the small intestine by stimulating the activity of a specific calcium-binding protein (CaBP) in its wall.
The normal level of parathyroid hormone is on average 0.15-0.6 ng/ml. It varies depending on age and gender. The average content of parathyroid hormone in the blood of people aged 20-29 years is (0.245±0.017) ng/ml, 80-89 years - (0.545±0.048) ng/ml; the level of parathyroid hormone in 70-year-old women is (0.728±0.051) ng/ml, in men of the same age - (0.466±0.40) ng/ml. Thus, the content of parathyroid hormone increases with age, but to a greater extent in women.
As a rule, several different tests should be used for the differential diagnosis of hypercalcemia.
We present the clinical and pathogenetic classification developed by us, based on the classification of O. V. Nikolaev and V. N. Tarkaeva (1974).
Clinical and pathogenetic classification of diseases associated with impaired secretion of parathyroid hormone and sensitivity to it
Primary hyperparathyroidism
- By pathogenesis:
- hyperfunctioning adenoma(s);
- hyperplasia of the OGD;
- hyperfunctioning parathyroid carcinoma;
- multiple endocrine neoplasia type I with hyperparathyroidism (Wermer's syndrome);
- multiple endocrine neoplasia type II with hyperparathyroidism (Sipple syndrome).
- According to clinical features:
- bone form:
- osteoporotic,
- fibrocystic osteitis,
- "Pagetoid";
- visceropathic form:
- with predominant damage to the kidneys, gastrointestinal tract, neuropsychiatric sphere;
- mixed form.
- bone form:
- Downstream:
- spicy;
- chronic.
Secondary hyperparathyroidism (secondary hyperfunction and hyperplasia of the parathyroid glands with prolonged hypocalcemia and hyperphosphatemia)
- Renal pathology:
- chronic renal failure;
- tubulopathy (Albright-Fanconi type);
- renal rickets.
- Intestinal pathology:
- malabsorption syndrome.
- Bone pathology:
- senile osteomalacia;
- puerperal;
- idiopathic;
- Paget's disease.
- Vitamin D Deficiency:
- kidney disease;
- liver;
- hereditary enzyme deficiencies.
- Malignant diseases: myeloma.
Tertiary hyperparathyroidism
- Autonomously functioning adenoma(s) of the parathyroid glands, developing against the background of long-standing secondary hyperparathyroidism.
Pseudohyperparathyroidism
- Production of parathyroid hormone by tumors of non-parathyroid origin.
Hormonally inactive cystic and tumor formations of the parathyroid glands
- Cyst.
- Hormonally inactive tumors or carcinoma.
Hypoparathyroidism
- Congenital underdevelopment or absence of the parathyroid glands.
- Idiopathic, autoimmune origin.
- Postoperative, developed in connection with the removal of the parathyroid glands.
- Postoperative due to impaired blood supply and innervation.
- Radiation damage, exogenous and endogenous (external beam radiation therapy, treatment of thyroid disease with radioactive iodine).
- Damage to the parathyroid glands due to hemorrhage or infarction.
- Infectious lesions.
Pseudohypoparathyroidism
- Type I - insensitivity of target organs to parathyroid hormone, dependent on adenylate cyclase;
- Type II - insensitivity of target organs to parathyroid hormone, independent of adenylate cyclase, possibly of autoimmune origin.
Pseudopseudohypoparathyroidism
The presence of somatic signs of pseudohypoparathyroidism in healthy relatives in families of patients with pseudohypoparathyroidism without characteristic biochemical disorders and without tetany.
 [ 13 ], [ 14 ], [ 15 ], [ 16 ], [ 17 ], [ 18 ], [ 19 ], [ 20 ], [ 21 ]
[ 13 ], [ 14 ], [ 15 ], [ 16 ], [ 17 ], [ 18 ], [ 19 ], [ 20 ], [ 21 ]

