Protein responsible for genetic inflammatory disease identified
Last reviewed: 14.06.2024

All iLive content is medically reviewed or fact checked to ensure as much factual accuracy as possible.
We have strict sourcing guidelines and only link to reputable media sites, academic research institutions and, whenever possible, medically peer reviewed studies. Note that the numbers in parentheses ([1], [2], etc.) are clickable links to these studies.
If you feel that any of our content is inaccurate, out-of-date, or otherwise questionable, please select it and press Ctrl + Enter.
A team of researchers led by Dr. Hirotsugu Oda from the CECAD Cluster of Excellence for Aging Research at the University of Cologne has discovered the role that a specific protein complex plays in some forms of immune dysregulation. This result may lead to the development of new therapeutic approaches aimed at reducing autoinflation and “restoring” the immune system of patients suffering from genetic dysfunction of this protein complex.
The study “Biallelic human SHARPIN loss of function induces autoinflammation and immunodeficiency” was published in Nature Immunology.
The linear ubiquitin-assembling complex (LUBAC), composed of the proteins HOIP, HOIL-1, and SHARPIN, has long been recognized for its critical role in maintaining immune homeostasis. Previous studies in mice have shown severe consequences of loss of SHARPIN, leading to severe dermatitis due to excessive skin cell death. However, the specific consequences of SHARPIN deficiency on human health have so far remained unclear.
The research team reports for the first time two humans with SHARPIN deficiency who exhibit symptoms of autoinflation and immunodeficiency, but unexpectedly do not exhibit dermatological problems, as seen in mice.
Upon further investigation, these individuals were found to have an impaired canonical NF-κB response, a pathway important for immune response. They also had increased sensitivity to cell death caused by members of the tumor necrosis factor (TNF) superfamily. Treatment of one of the patients with SHARPIN deficiency with anti-TNF therapy, which specifically inhibits TNF-induced cell death, resulted in complete resolution of autoinflation at the cellular level and in clinical presentation.
Research shows that excessive and uncontrolled cell death plays a critical role in human genetic inflammatory diseases. Oda's team has added SHARPIN deficiency as a new member of a group of genetic human inflammatory diseases that they propose to call "inborn errors of cell death."
Protection from Immune Dysregulation The study was initiated in the laboratory of Dr. Dan Kastner at the National Institutes of Health (NIH) in the United States. Scientists there were able to observe one patient with childhood onset of unexplained episodes of fever, arthritis, colitis and immunodeficiency.
After obtaining informed consent, they performed exome sequencing on the patient and his family and discovered that the patient had a disruptive genetic variant in the SHARPIN gene leading to undetectable levels of SHARPIN protein. They also found that patient cells showed an increased propensity to die in both cultured cells and patient biopsies.
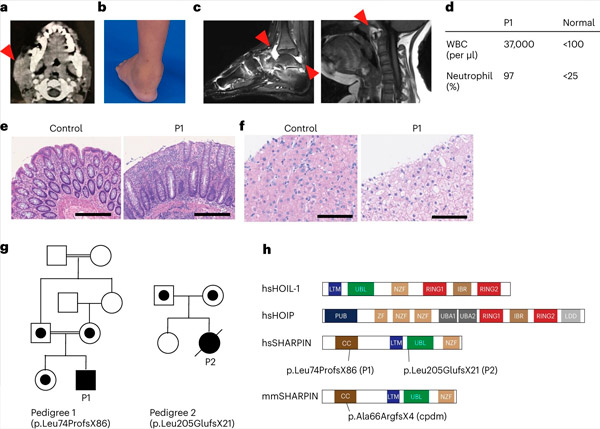
SHARPIN deficiency in humans causes autoinflammation and liver glycogenosis. Source: Nature Immunology (2024). DOI: 10.1038/s41590-024-01817-w
The team also found that the development of lymphoid germinal centers - specialized microstructures in the adenoids that are critical for the maturation of our immune system's B cells and therefore the production of antibodies - was significantly reduced due to increased B cell death. These results explain the immunodeficiency in patients and highlight the important role of LUBAC in maintaining immune homeostasis in humans.
“Our study highlights the critical importance of LUBAC in protecting against immune dysregulation. By elucidating the molecular mechanisms underlying LUBAC deficiency, we are paving the way for new therapeutic strategies aimed at restoring immune homeostasis,” said Oda, lead author of the study.
He added: “One of the patients with SHARPIN deficiency had been wheelchair dependent for many years before we first saw him. His ankles were sore and it was too painful to walk. The genetic diagnosis allowed us to target the correct molecular pathway underlying his conditions."
Since the patient began receiving anti-TNF therapy, he has been symptom-free for almost seven years. “As a clinician and scientist, I am pleased to have the opportunity to positively impact one patient's life through our research,” Oda concluded.
