Thalidomide derivatives lead to the death of resistant cancer cells
Last reviewed: 14.06.2024

All iLive content is medically reviewed or fact checked to ensure as much factual accuracy as possible.
We have strict sourcing guidelines and only link to reputable media sites, academic research institutions and, whenever possible, medically peer reviewed studies. Note that the numbers in parentheses ([1], [2], etc.) are clickable links to these studies.
If you feel that any of our content is inaccurate, out-of-date, or otherwise questionable, please select it and press Ctrl + Enter.
A study conducted by Goethe-University Frankfurt points to the possibility that thalidomide derivatives are potentially suitable for treating cancer. In the 1950s, thalidomide was sold as a sleeping pill. It later became notorious for causing serious malformations in the fetus in the early stages of pregnancy.
The molecule is also known to mark proteins in the cell for their destruction. As part of the current research, scientists created derivatives of thalidomide. They were able to show that these substances affect the destruction of proteins responsible for the survival of cancer cells.
Perhaps no other molecule has such a turbulent past as thalidomide. It was the main component of a drug approved in many countries in the 1950s as a sedative-hypnotic. However, it soon became clear that pregnant women taking thalidomide often gave birth to children with serious deformities.
However, in recent decades, medicine has again placed high hopes on it. Research has shown, among other things, that it inhibits the growth of blood vessels and is therefore potentially suitable for cutting off tumors from their nutrient medium. Then it also proved to be very effective in the treatment of multiple myeloma, malignant tumors in the bone marrow.
“We now know that thalidomide can be called a 'molecular glue',” explains Dr. Xinglai Cheng from the Institute of Pharmaceutical Chemistry at Goethe-University Frankfurt. "This means that it is able to grab two proteins and join them."
This is especially interesting because one of these proteins is a kind of "labeling machine": it attaches an unambiguous "TRASH" label to another protein.
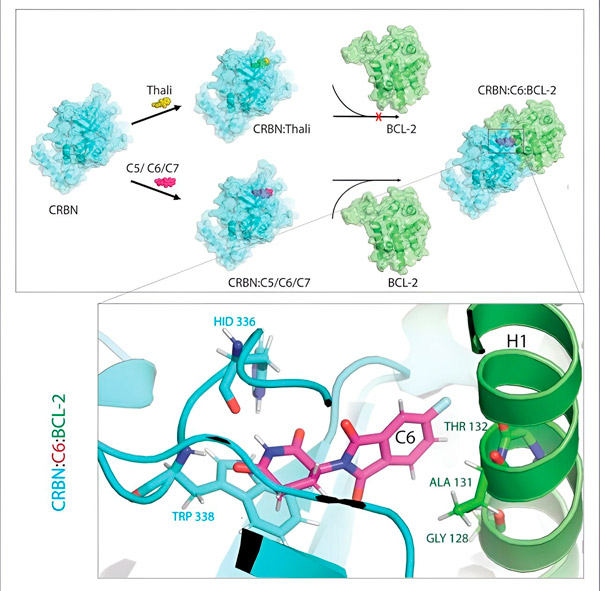
Thalidomide derivatives C5, C6 and C7 modify CRBN, the "labeling machine", so that it can bind to BCL-2. Thus, the BCL-2 molecule is marked for degradation—a possible new strategy to fight cancer. Author: Dr. Xinglai Cheng
The cell's waste disposal system recognizes this tag: it grabs the labeled protein molecule and shreds it. "It is this mechanism that explains the different effects of thalidomide," says Cheng. "Depending on which protein is tagged, this can lead to deformities during embryonic development or to the destruction of malignant cells."
This mechanism offers great medical promise because cancer cells depend on certain proteins to survive. If they could be systematically targeted and shredded, the disease might be cured. The problem is that molecular glue is quite peculiar.
One of its binding partners is always a cell marking machine, or in scientific parlance, an E3 ligase called CRBN. Only a very few of the many thousands of proteins in the body can be a second partner - which ones depend on the glue.
"So we created a series of thalidomide derivatives," says Cheng. "We then investigated whether they had adhesive properties and, if so, what proteins they were effective against." To do this, the researchers added their derivatives to all the proteins in the grown cell line. They then observed which of these proteins then degraded in the presence of CRBN.
"In the process, we identified three derivatives that could tag a cellular protein that is very important for degradation, BCL-2," explains Cheng. "BCL-2 prevents the cells from activating their self-destruction program, so if it's missing, the cells die."
BCL-2 has therefore long been the focus of cancer research. There is even already a drug for leukemia called venetoclax, which reduces the effectiveness of BCL-2 and thus causes the mutated cells to self-destruct.
"However, in many cancer cells, BCL-2 itself is mutated. As a result, venetoclax no longer inhibits the protein," says Cheng. "We were able to show that our derivatives also mark this mutated form for degradation. In addition, our partners at the Max Planck Institute for Biophysics simulated the interaction of thalidomide derivatives with BCL-2 on the computer. This showed that the derivatives bind to completely different sites than venetoclax - a result that we were later also able to confirm experimentally."
In addition, the researchers tested their substances on fruit flies with cancer cells. The survival rate of flies treated in this way was significantly higher. However, Cheng cautions against getting too excited because these results are still basic research. "Although they show that the modified thalidomide molecules have great therapeutic potential, we cannot yet say whether they will prove themselves in practice at any point in time."
The study results were published in the journal Cell Reports Physical Science.
